Imaging Findings - Pancreatic Parenchymal
- Should be considered in the context of clinical and pathological findings
- Considerable similarity in appearances compared with other differential diagnoses (carcinoma,
lymphoma)
- Findings depend upon degree of inflammation and resulting fibrosis
- Multimodality approach may be required including CT,
MRI,
ultrasound (transabdominal and endoscopic),
ERCP and FDG PET/CT more recently
Transabdominal Ultrasound
- Often the initial imaging modality
- Non-specific appearances
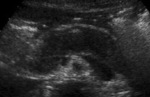
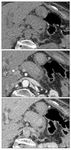
- Similar to acute pancreatitis features - namely diffusely enlarged hypoechoic pancreas (Fig.
16)
Endoscopic Ultrasound
- Significant role in diagnosis of AIP
- EUS-guided biopsy
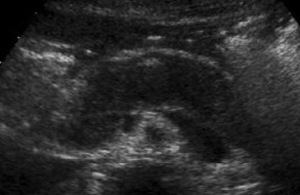
Fig. 16: US image of 45 year old male with AIP showing diffuse enlargement of the pancreas
References: Morana et al. (2005) Autoimmune pancreatitis: instrumental diagnosis. Journal of pancreas online 28-30
CT imaging - Pancreatic Parenchymal
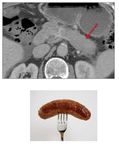
Diffuse or focal pancreatic enlargement with loss of the normal lobular architecture "sausage-shaped" pancreas found in 40-60% of cases 12 (Fig.
17)
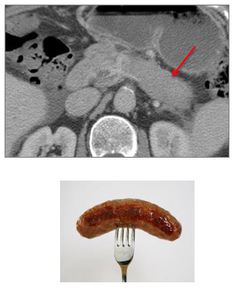
Fig. 17: CT demonstrating "Sausage-shaped" pancreas
- Hypoattenuating/intense peripheral rim
- "Capsule or Envelope" (12-40%)
- "Halo Sign"
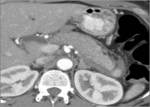
- Thickening of the pancreatic "envelope" (Fig.
18)
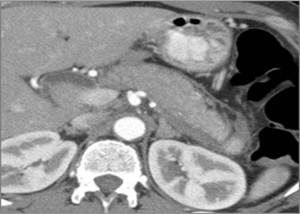
Fig. 18: CECT demonstrating thickening of the pancreatic envelope
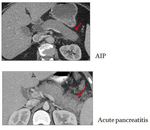
- Minimal peripancreatic fat stranding when compared with acute pancreatitis seen on CT (Fig.
19)
- Fluid collections and pseudocysts are less likely
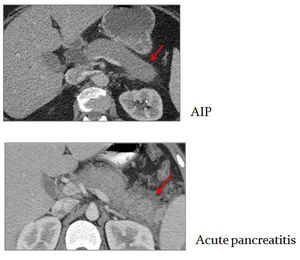
Fig. 19: CECT appearances of AIP vs Acute pancreatitis fat stranding
- Altered enhancement pattern (Fig.
20)
- Reduced during pancreatic phase
- Increased during PV phase 13
- Delayed pattern (90% cases 14)
- Important for differential diagnosis
- Features vary depending on degree of fibrosis/inflammation/length of disease
- Calicification and parenchymal atrophy uncommon
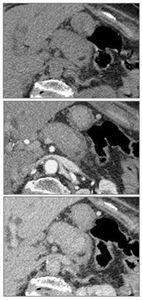
Fig. 20: Pre-, pancreatic parenchymal and delayed phase CECT demonstrating delayed pancreatic enhancement in AIP
MR Imaging - Pancreatic Panrenchymal
- Low signal pancreas on T2-weighted imaging
- Peripheral "capsule" of low attenuation/signal seen on both CT and MRI in 12-40% of cases thought to represent inflammatory changes in the peripancreatic tissues 15 (Fig.
21)
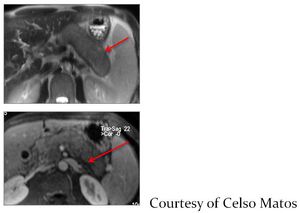
Fig. 21: MRI demonstrating inflammatory peripancreatic "capsule"
References: Prof. Celso Matos, Hospital Erasme, Brussels, Belgium
Imaging Findings - Pancreatic Ductal
- Irregular narrowing main pancreatic duct (Figs.
22&23) greater than one third of the length of the duct
- Relative lack of duct dilatation proximal to stricture
- Loss of normal duct branching structure also recognised 16 but presence of side branches at the portion of narrowed pancreatic duct are seen in AIP
- Different patterns depend on degree of pancreatic involvement
- MRCP may demonstrate diffuse narrowing of the main pancreatic duct 17
- Ductal imaging modalities used varies with geography,
for example in Europe/USA MRCP is frequently used to reduce possible complications of invasive ERP,
commonly used in Asia
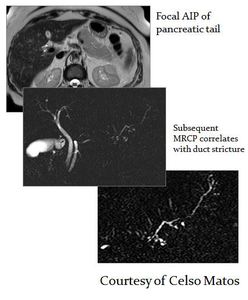
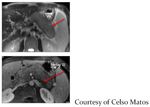
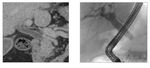
Fig. 22: MR findings of pancreatic ductal AIP
References: Prof. Celso Matos, Hospital Erasme, Brussels, Belgium
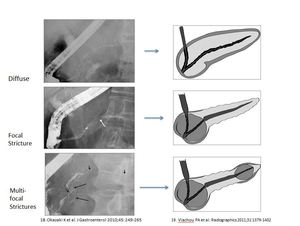
Fig. 23: Spectrum of AIP ductal involvement on ERP with accompanying schematic representation
References: Okazaki K et al. (2010) J Gastroenterol 45:249-65 and Vlachou PA et al. (2011) Radiographics 31:1379-1402
FDG PET/CT Imaging
- Provides information about disease activity in staging,
guiding biopsy and monitoring response to treatment but no established role in AIP
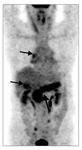
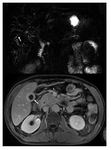
- FDG uptake in AIP reflects sites of organ involvement (diffuse/focal) (Fig.
24)
- Imaging appearances overlap with pancreatic adenocarcinoma and lymphoma
- The use of Fluorine-18 fluoroarabinofuranosyl cytosine (FAC) as alternative tracer to FDG may be sensitive to localised immune reaction and may therefore offer new insight into autoimmune disorders 20
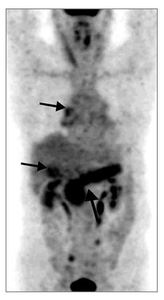
Fig. 24: PET/CT demonstrating FDG uptake in organs involved in IgG4-related disease
Extra-Pancreatic Manifestations of Type 1 AIP - Biliary System
- Commonest extrapancreatic site of AIP involvement manifesting as IgG4-related sclerosing cholangitis 21
- Focal or diffuse mural thickening,
stenoses,
luminal irregularity and increased post contrast enhancement 22
- Most commonly effects the intrapancreatic section of the common bile duct which leads to proximal bile duct dilatation and obstructive jaundice (Fig.
25)
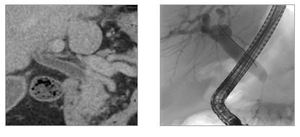
Fig. 25: CBD enhancement on CECT and intrapancreatic biliary duct stricture on ERCP
- Best demonstrated on MRCP
- Appearances can mimic ductal cholangiocarcinoma particularly when associated with a soft tissue mass 23
- May be difficult to differentiate IgG4-related sclerosing cholangitis from primary sclerosing cholangitis (Fig.
26)
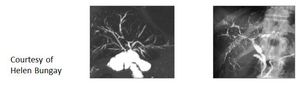
Fig. 26: Multi focal strictures of the biliary tree in IgG4-related sclerosing cholangitis seen on MR and ERCP
References: Dr. Helen Bungay, Department of Radiology, Oxford University Hospitals Trust/England 2014
- Gallbladder involvement has also been reported
- Diffuse gallbladder wall thickening secondary to inflitration with IgG4 plasma cells and resulting fibrosis
- Hypoechoic thickened wall on US
- Hypointense gallbladder wall on T2 weighted MRI with contrast enhancement which persists on delayed phase images 23
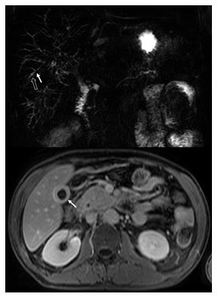
Fig. 27: MRI: IgG4-related sclerosing cholangitis
CT: Thickened enhancing gallbladder wall
References: Tan TJ et al. (2014) Extrapancreatic findings of IgG4-related disease. Clin radiol 69:209-18
Differentiation from Pancreatic Cancer
- Pancreatic carcinoma is the most important of the differential diagnoses of AIP to consider
- Both may appear as a focal pancreatic mass and can therefore be challenging to differentiate
Some features to aid diagnosis of focal AIP include:
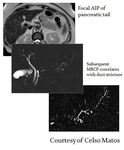
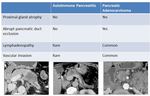
Imaging characteristics (Fig.28):
- Delayed enhancement in hepatic phase on CECT - greater than that seen in pancreatic carcinoma 13
- Tapered pancreatic duct occlusion in AIP compared with abrupt oclusion in pancreatic cancer
- No atrophy of the proximal gland
- Unlikely lymphadenopathy
- No vascular invasion as seen with pancreatic cancer
- Presence of other organ involvement is more suggestive of AIP
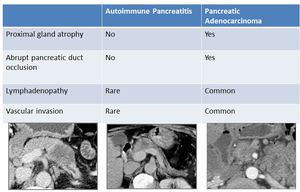
Fig. 28: Features to differentiate focal AIP from pancreatic cancer
- Kamisawa et al suggested DWI is useful for detecting AIP and as a biomarker for evaluting response to steroids 24
- Both AIP and pancreatic cancer are seen as high signal areas on DWI but these areas in AIP are differently shaped,
namely diffuse,
solitary or multifocal compared with the solitary areas seen in pancreatic cancer25
- ADC values significantly lower in AIP compared with pancreatic cancer
- Early work on Receiver operating characteristic (ROC) analysis suggests an optimal ADC cut off of 1.075 x 10-3mm2/s to distinguish AIP from pancreatic cancer24 (Fig.
29)

Fig. 29: DWI and ADC map images demonstrating restricted diffusion of AIP within distal pancreas (ADC value less than that seen in pancreatic cancer)
References: Kamisawa et al. (2010) Differentiation of autoimmune pancreatitis from pancreatic cancer by diffusion weighted MRI. Am J Gastroenterol 105:1870-5
Clinical/Biochemical characteristics:
- Response to steroids highly suggestive of AIP
- Diagnostic accuracy in differentiating focal AIP from pancreatic cancer can be further improved with combined serum CA19-9 (<85U/ml) and IgG4 levels (>280mg/dl) (IgG4 may not be raised in Type 2 AIP) as demonstrated by Chong et al26
It should therefore be noted that a careful mulitdisciplinary approach,
considering the features detailed above should be employed to diagnose focal AIP.
Review of previous imaging and histology and correlation with clinical parameters is vital.
Treatment Options/Advances
- Dramatic steroid response in AIP regardless of subtype
- Response to steroids can be both therapeutic and diagnostic (once pancreatic cancer has been ruled out,
a two week steroid trial could be commenced in the absence of typical imaging/biochemical features)
- Starting dose for inducting remission 0.6-1 mg/kg per day
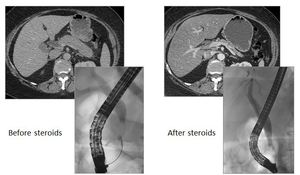
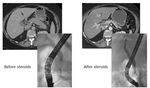
Fig. 30: CT/ERCP pictures of AIP pre and post steroid treatment demonstrating resolution of AIP imaging features
- Response to steroids is based upon objective data such a radiologic evidence of dramatic improvement in appearances (Fig.
30),
resolution of obstructive jaundice without biliary stenting and normalization of liver function tests25
- Most severe cases of AIP (i.e.
relapsing,
poorly controlled disease) may require surgical intervention
Current Treatment of AIP Type 1 - Initial Attack:

Fig. 31: Treatment algorithm for Type 1 AIP: Initial attack
References: Kamisawa T, Shimosegawa T, Okazaki K, Nishino T, Watanabe H, Kanno A et al. (2009) Standard steroid treatment for autoimmune pancreatitis. Gut 58: 1504-7
Current Treatment of AIP Type 1 - Relapse:

Fig. 32: Treatment algorithm for Type 1 AIP: Relapse
References: Kamisawa T, Shimosegawa T, Okazaki K, Nishino T, Watanabe H, Kanno A et al. (2009) Standard steroid treatment for autoimmune pancreatitis. Gut 58:1504-7